Dr. Elan Ziv in Brussels to present revolutionary device that improves quality of life for millions of women
Statistics show that over 50% of women worldwide experience either stress urinary incontinence, pelvic organ prolapse, or bowel incontinence. In the case of pelvic organ prolapse alone, it is estimated that around 135 million women in the world suffer this condition, out of which nearly 30% of the occurrences happen in Europe. Pelvic organ prolapse (POP) happens when the muscles and tissues supporting the pelvic organs (uterus, bladder, rectum) become weak. Because of their weight, pelvic organs might create pressure downwards, into the vagina, and potentially become exposed.
“It’s a topic that has become a taboo for many women,” said Dr. Elan Ziv, CEO and Medical Director at ConTIPI, who came to Brussels to raise awareness about these conditions and present a solution. “If you think about it, suffering from any of these forms of incontinence is difficult to talk about. And not only that, organ prolapse affects the sexual lives of so many women.”

To support the pelvic floor two alternatives have been commonly used. Either a surgery, in which the results are unsatisfactory due to the high rate of adverse events – more than 40% of patients who undergo surgery see a relapse. Or the second option, the current non-invasive mode e.g. using a pessary: a reusable silicone device that is inserted in most cases by a doctor into the vagina. It can be kept inside up to 3 months before being taken out, cleaned and reinserted for another 3 months. This process is often mired with discomfort, discharge and infections, as well as the inability to have intercourse.
“There has not been any innovation in this area for 23 years,” said Dr. Ziv. “And this is a medical condition that affects the quality of life of millions of women all over the world. And that is why we developed this product. A product that can be used by any woman afflicted by pelvic organ prolapse. They can do it in the comfort of their home. Women would no longer need to go to the doctor to have the device inserted or removed. And this is, for me, the highest value of the product: giving women agency over when to use it, and when to remove it.”
ConTIPI has designed a disposable vaginal device which is inserted into the vagina with an easy-to-use applicator. This is done by the user herself, not a medical practitioner. The device may be kept inside the body for up to 7 days, after which the user pulls a string and the device shrinks back to small dimensions, reducing the discomfort to a minimum. The device is then discarded along with the applicator.
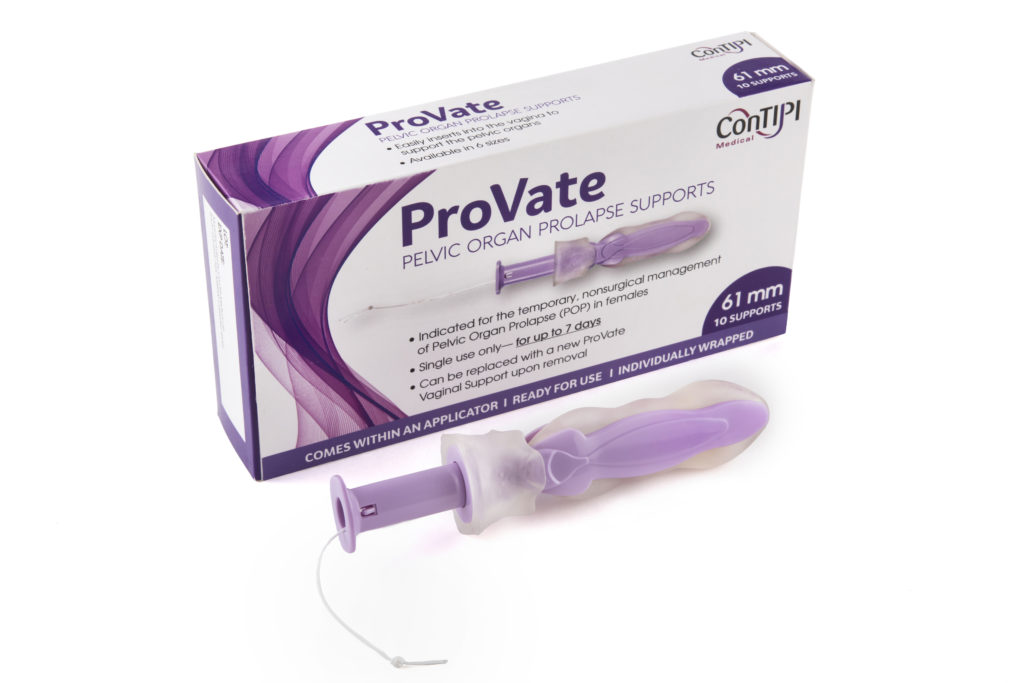
“The idea is that the patient goes to the doctor only one time, the very first time, and gets the prescription,” said Dr. Ziv. “From then on the patient can buy it whenever she needs to. There’ll come a time when all she needs to do is order it online.”
This is the second product designed by ConTIPI in FemTech, a high-tech category in women’s health arena. The intellectual property of the first device, which was designed for stress urinary incontinence, was acquired by Kimberly Clark Worldwide and is already available on pharmacy shelves in North America. Their device for POP is ready for the market. “We are really excited to share the news that our product has received the 510(k) clearance from the Food and Drug Administration for marketing in the US, as well as the CE marking in Europe.”
In the US, the current solution for POP –the pessary– has a cost per patient of USD 420 (EUR 377) to the national health service. For ConTIPI’s new product, Dr. Ziv estimates that the cost of the device will be the same, USD 420, considering a supply of 120 devices a year per patient. “That’s about USD 3.50 (EUR 3.14) per device,” said Dr. Ziv.
As it was the case with their first device and its subsequent sale to Kimberly Clark Worldwide, ConTIPI is open to collaborate with other companies to take its new product to market and reach its full potential. “We are a growing company. We are looking for the right firms with whom we can create partnerships.”
When asked about the environmental side effects of having both a disposable plastic device and an applicator, Dr. Ziv acknowledged that this was one of the main challenges. “Yes, true, that is an issue, the plastic. We’ve thought about it a lot. You will agree that this is a discussion that needs to involve the patient as well because if we produce the device with bio-degradable materials the cost would go up to about USD 10 (EUR 8,97). Are they going to be willing to pay the premium? Hopefully the answer would be yes. And hopefully in the near future we will be able to produce the device with bio-degradable materials that match the current costs.”